The SfRBM Foundation was designed to foster and support SfRBM’s education, training, and research programs as well as fund new developments and opportunities in these areas.
In fact, since 1995, SfRBM has provided over $450,000 in Research Mini-Fellowships, Travel, and Young Investigator Awards to support scientists in early stages of their careers.

There are a number of ways to contribute, including one-time donations and Planned Giving. As 2019 comes to a close, please consider including the SfRBM Foundation in your giving plans.
Here are 5 reasons to donate to the SfRBM Foundation Today:
Please call us today at (317) 205-9482 to learn more about Planned Giving Opportunities. 
— Published
Categories: Free Radical Biology and Medicine, Education, SfRBM Trainee Council, Research, Redox Biology
By: Archit Rastogi, Ph.D. Candidate, Molecular & Cellular Biology Graduate Program, University of Massachusetts, Amherst
Oxygen and reactive oxygen species play fundamental roles in biology, both directly and indirectly. Many transcriptional and translational changes occur as a direct result of cellular oxygen concentrations. Yet, all life did not evolve with oxygen on the planet, the Earth’s atmosphere only became oxygenated 2.4 billion years ago. Thus, high oxygen concentrations are an evolutionary selective pressure for all organisms that evolved since. Higher intracellular concentrations of oxygen lead to free radical generation, destabilizing cell membranes and causing cell death. To survive the duality of oxygen – it is both essential to life and toxic to life – organisms have evolved various redox defenses. While many understand the role of oxygen in producing cellular energy, ATP, important research is also conducted on the anti-oxidant defenses of cells and organisms. The current research focuses on glutathione, an intracellular anti-oxidant molecule that neutralizes free radicals. 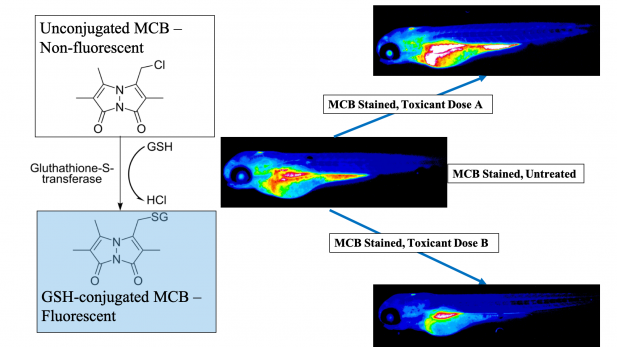
Glutathione is the predominant vertebrate redox buffer present in intracellular concentrations in the millimolar range. This study hypothesized that glutathione levels are tightly regulated throughout development and, disruption of these levels would lead to adverse health outcomes later in life. Understanding the role of glutathione during development can also help explain the evolutionary adaptations that promoted life on earth after oxygenation. During embryogenesis, glutathione protects developing organs from oxidative and toxic insults. Glutathione levels change in a specific, directional pattern conserved across vertebrates; these changes are linked to key developmental events and disruptions lead to altered cell fate decisions and cell death. Different organs are known to have diverging redox resiliencies, especially during development. However, owing to technical limitations, limited data about glutathione dynamics in a whole embryo exist.
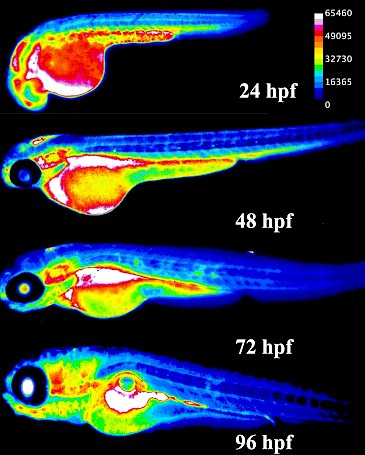
Glutathione utilization is tightly spatiotemporally controlled during vertebrate embryonic development.
This study was recently conducted as part of my doctoral dissertation at the University of Massachusetts, Amherst and demonstrates the spatiotemporal changes that occur in glutathione levels during vertebrate embryogenesis in vivo. We utilized monochlorobimane, a dye that forms a fluorescent adduct with reduced glutathione to visualize these changes. Using small molecule modulators of the glutathione pathway, we established the specificity and sensitivity of this method in the zebrafish (Danio rerio) embryo. Zebrafish serve as an ideal model species for this study, owing to their high fecundity and rapid exogenous development. Moreover, their redox biology closely mirrors humans – our data align with glutathione levels during human embryonic development.
Read the full study here in Redox Biology, an official journal of SfRBM: https://www.sciencedirect.com/science/article/pii/S2213231719302757
The organ-specific glutathione localization patterns we found lay the groundwork for determining discrete redox-sensitive temporal windows during embryogenesis. As illustrative examples of this, we tested two environmental toxicants – PFOS (a surfactant in non-stick cookware and firefighting foam) and MEHP (a bioactive metabolite of a ubiquitous plasticizer). We found that monochlorobimane responded in a dose-dependent, age and structure specific manner to both chemicals, indicating its broad application potential as an unbiased method to assess glutathione interruptions arising from developmental chemical exposures in live embryos.
— Published
By: Alexander Michels, Ph.D., Oregon State University
The 2019 Linus Pauling Institute 10th International Conference and SfRBM Regional Symposium will be a special event. For the first time, this year’s conference will be undertaken in conjunction with SfRBM.
Given the strong ties between the SfRBM and the Linus Pauling Institute for the last few decades, it is a fitting relationship between our two organizations. The Conference will be held August 14-16, 2019 at Oregon State University in Corvallis, Oregon.
The scientific sessions of the conference – Bioactives, Botanicals, and Redox Mechanisms - will encompass new approaches and therapies in neurodegenerative conditions, the role of redox biology in neurobiology and cancer, and the role that dietary supplements can play in health.
Every session in the conference touches one or more of these areas, and many speakers bridge divides between these fields. The hope of this event is to bring scientists and clinicians of diverse backgrounds and interests together; to find ways we can work together and break new ground in scientific discovery.
While the Linus Pauling Institute’s mission is research-based, it also includes a commitment to scientific outreach. Therefore, every conference we create has a session dedicated to the dissemination of scientific concepts and practical information gleaned from scientific studies to the public.
This year, our conference will close with a keynote address from Dr. Louis J. Ignarro. Dr. Ignarro, along with Ferid Murad and Robert Furchgott, received the Nobel Prize in Physiology or Medicine in 1998 for elucidating the biological roles of nitric oxide.

In the modern era of redox biology, it is difficult to think of a time where the properties of a ubiquitous free radical such as nitric oxide were unknown. When Dr. Ignarro started to work on the effects of nitric oxide in the body, it was certainly not a new topic - Alfred Nobel himself worked with nitroglycerin (although more on its explosive properties). In Nobel’s day, nitroglycerin was already being used for the relief of angina symptoms by promoting blood vessel relaxation – even though nobody knew how it worked.
In the late seventies, Ferid Murad showed how nitroglycerin activated enzymes that caused blood vessels to expand, and it has this effect through the production of nitric oxide. Louis Ignarro expanded on this work and began a search for a substance produced in the innermost layer of blood vessels that produced a similar effect. Ignarro and Furchgott, independently – yet simultaneously – revealed that the body did indeed produce its own nitric oxide.
The discovery has made possible many new medications, such as those used to treat heart and cardiovascular diseases. It was also the groundbreaking discovery that led to an all-too-well-known blue pill for the treatment of impotence.
Dr. Ignarro’s keynote lecture, The Road to Stockholm – A Nobel Mission will explore his journey to the Novel prize, but it will also focus on his humble beginnings. The Brooklyn-born son of Italian immigrants, Ignarro attributes his early life with much of his later success, including a chance encounter with Dr. Linus Pauling. In his own words:
“I first met Linus Pauling in 1956, two years after [he received] his Nobel Prize in Chemistry. My high school chemistry teacher invited him to Long Beach High to assist in setting up a chemistry lab to complement the course lectures, and also to give a general experimental demonstration to the high school in the auditorium.
Since my teacher knew of my passion for chemistry, I had the opportunity to work with Professor Pauling for many hours. There has never been any doubt in my mind that Linus Pauling was the most important motivating factor accounting for my success as a chemical pharmacologist.”
Among his many scientific awards, Dr. Ignarro was elected to the National Academic of Sciences, the American Academy of Arts and Sciences, and the National Academy of Medicine. Dr. Ignarro is currently Professor Emeritus, Department of Molecular and Medical Pharmacology at UCLA.
The Linus Pauling Institute International Conference, SfRBM regional symposia, and International Society of Neurochemistry symposia, will be held August 14-16, 2019, in Corvallis, Oregon. Discounted registration is available for all SfRBM members.
— Published
By: Phyllis A. Dennery, MD
How do physicians differ from basic scientists in their approach to diseases? The former provide insights into disease processes and their manifestations whereas, the latter delve deeply into mechanisms and look for potential therapeutic approaches. If we capitalize on these differences, this could lead to an ideal partnership between those who witness disease at the bedside and those who can adeptly use ever-evolving tools to elucidate the mechanistic underpinnings of human pathology.
 Why is this partnership not always successful? Communication is often challenging due to limited understanding of each participants perspective. In fact, the gap is widening between the language spoken by biomedical scientists and clinicians/physician-scientists. With exponential advances in molecular biology and genetics, it has become difficult to incorporate these novel understandings into everyday medical practice. Philosophically, the two groups have different goals. Science for the sake of science is laudable and necessary to organically discover new insights. However, the pressures of finding a solution for diseases exist and are much more vivid for the physician-scientists who want solutions for their patients. Clinicians want to save lives and have immediate impact but are hampered by the fear of injury and/or bad outcomes. Scientists are encouraged to challenge paradigms and to develop new approaches which leads to a new found knowledge without the fear of such consequences. There needs to be a way to bridge the divide between the culture of basic scientists and that of physician-scientists.
Why is this partnership not always successful? Communication is often challenging due to limited understanding of each participants perspective. In fact, the gap is widening between the language spoken by biomedical scientists and clinicians/physician-scientists. With exponential advances in molecular biology and genetics, it has become difficult to incorporate these novel understandings into everyday medical practice. Philosophically, the two groups have different goals. Science for the sake of science is laudable and necessary to organically discover new insights. However, the pressures of finding a solution for diseases exist and are much more vivid for the physician-scientists who want solutions for their patients. Clinicians want to save lives and have immediate impact but are hampered by the fear of injury and/or bad outcomes. Scientists are encouraged to challenge paradigms and to develop new approaches which leads to a new found knowledge without the fear of such consequences. There needs to be a way to bridge the divide between the culture of basic scientists and that of physician-scientists.
Of course, the integration of newfound knowledge of basic science insights into clinical decision making is important to improve patient care. However, once a discovery is made, dissemination and adoption in clinical practice is another significant hurdle which requires community engagement and public health support. This path is riddled with pitfalls. Therefore, it is often difficult to see how certain basic concepts lead to therapeutic interventions and to clinical successes. This has been a big challenge for redox biology in that despite years of clear ascertainment that antioxidants are beneficial to prevent cytotoxicity in various model systems, the applications to large scale human trials have been extremely limited and discouraging. This is likely due to the fact that the human organism is much more complex than any other laboratory-based model and there are many more factors that come into play to alter outcomes, in particular, disparities in healthcare delivery and lack of cultural competence. Nevertheless, a partnership between physicians and scientists may overcome these hurdles by identifying ways to enhance a particular benefit without causing harm.
Recently, we have seen some encouraging success stories around treatment of disease using redox biology concepts. Hopefully, this is the beginning of a new era. The process from discovery to implementations is long and tortuous. A recent publication reviewed the literature and identified papers where a scientific discovery was made followed by its adaptation into healthcare. On the average, it took 17 years for this to happen (1). This means that we have to tolerate the uncertainty between basic discoveries and eventual adaptation into clinical practice for many years and not lose faith. This is not for the faint of heart and requires constant vigilance to continue to focus on the ultimate goal through partnership and collaboration.
Many will argue that we have to conduct science for the sake of science, rather than focus on finding an immediate solution to a disease process. I would say that we have to adopt both approaches. Families, patients, their families and communities rely on us to help improve their health. Without a goal towards therapeutic discoveries, we will extinguish their hopes and erode their faith in us.
How can we then work together as physician-scientists and basic scientists? We can be patient with each other in terms of understanding each other’s language. Oftentimes the physician rolls her eyes when PhD colleagues try to describe a disease process, smugly dismissing the limited understanding. In return, the basic scientist is exasperated by the approaches taken by the physician-scientist to solve a problem, and chastise him for his lack of rigor or his choice of vague assays. Working together and encouraging each other will go farther. Trying to formalize relationships between the most basic and most clinical investigators would make for an synergistic opportunity to accelerate discovery and implementation.
In the past 28 years, it has been my distinct pleasure to be engaged with SfRBM, known previously as the Society for Free Radical Biology and Medicine and The Oxygen Society. I have enjoyed every iteration of this Society and have felt compelled to be an active participant despite feeling inadequate in my lack of formal training in chemistry, biochemistry, molecular biology and genetics. I feel, nonetheless, that we physician-scientists can bring value to the Society in many ways.
As I approach the Presidency of the SfRBM as its first MD leader, I will look forward to working with you and learning from you to speak a common language that enhances scientific discovery and ultimately promotes health.
1. Morris ZS, Wooding S, Grant J. The answer is 17 years, what is the question: understanding time lags in translational research. J R Soc Med. 2011;104(12):510-20.
— Published
Categories: Free Radical Biology and Medicine, Redox Biology
Part 2 of 4 on “Exploring Careers in Industry” by SfRBM Nominations/Leadership Development Chair Anne Diers, Ph.D. (Dr. Diers on LinkedIn: https://www.linkedin.com/in/annerdiers/)
Looking for a job in academia versus elsewhere can be a very different process. LinkedIn is much more important in the non-academic setting. Make sure your LinkedIn page is current, professional, and thoughtful. You can also use LinkedIn to find out more about companies you're interested in, connect to people, network for informational interviews and/or positions (a personal introduction to a hiring manager can be very helpful), and look for position announcements.

Connect with SfRBM on LinkedIn at - https://www.linkedin.com/company/society-for-free-radical-biology-and-medicine/
Making the leap to industry can be very challenging. You need to target your search and be very active in it - your time will be better spent networking than just applying positions from the comforts of your home.
For the hiring manager, their biggest concern is that they’ll make the wrong decision on who to hire, ending up with someone who is not be able to accomplish the business objectives at hand. With that perspective, it becomes clear that the best way to get a job in industry is to allay those fears in the hiring manager. Or in other words, connect your skills to the needs listed in the job posting – be specific! That middle-author manuscript you’re on because you did a bit of confocal microscopy for the lab down the hall should become “demonstrated proficiency working in a collaborative team environment” on your resume. It also helps to have a personal introduction if possible. To do this, you’ll need to identify an opening, and then get in touch with someone who can help you get that introduction. Again, LinkedIn can help you get started; you can see who’s where and identify the connections you may not even know you already have. And don’t forget, you’ve got lots of networks to tap (not just those on LinkedIn) – including at SfRBM’s Annual Conference - because everybody knows somebody who knows somebody!
In part 3 of this series, Dr. Anne Diers will explore the topic “Resume ≠ Curriculum Vitae” In her series “Exploring Careers in Industry.”
— Published
Category: Education