By: Archit Rastogi, Ph.D. Candidate, Molecular & Cellular Biology Graduate Program, University of Massachusetts, Amherst
Oxygen and reactive oxygen species play fundamental roles in biology, both directly and indirectly. Many transcriptional and translational changes occur as a direct result of cellular oxygen concentrations. Yet, all life did not evolve with oxygen on the planet, the Earth’s atmosphere only became oxygenated 2.4 billion years ago. Thus, high oxygen concentrations are an evolutionary selective pressure for all organisms that evolved since. Higher intracellular concentrations of oxygen lead to free radical generation, destabilizing cell membranes and causing cell death. To survive the duality of oxygen – it is both essential to life and toxic to life – organisms have evolved various redox defenses. While many understand the role of oxygen in producing cellular energy, ATP, important research is also conducted on the anti-oxidant defenses of cells and organisms. The current research focuses on glutathione, an intracellular anti-oxidant molecule that neutralizes free radicals. 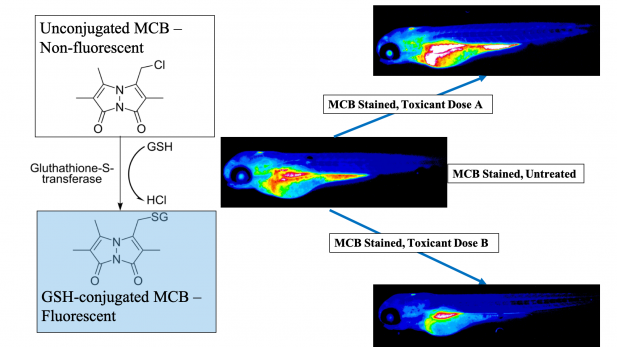
Glutathione is the predominant vertebrate redox buffer present in intracellular concentrations in the millimolar range. This study hypothesized that glutathione levels are tightly regulated throughout development and, disruption of these levels would lead to adverse health outcomes later in life. Understanding the role of glutathione during development can also help explain the evolutionary adaptations that promoted life on earth after oxygenation. During embryogenesis, glutathione protects developing organs from oxidative and toxic insults. Glutathione levels change in a specific, directional pattern conserved across vertebrates; these changes are linked to key developmental events and disruptions lead to altered cell fate decisions and cell death. Different organs are known to have diverging redox resiliencies, especially during development. However, owing to technical limitations, limited data about glutathione dynamics in a whole embryo exist.
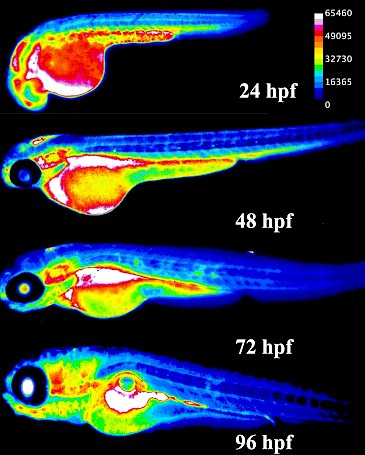
Glutathione utilization is tightly spatiotemporally controlled during vertebrate embryonic development.
This study was recently conducted as part of my doctoral dissertation at the University of Massachusetts, Amherst and demonstrates the spatiotemporal changes that occur in glutathione levels during vertebrate embryogenesis in vivo. We utilized monochlorobimane, a dye that forms a fluorescent adduct with reduced glutathione to visualize these changes. Using small molecule modulators of the glutathione pathway, we established the specificity and sensitivity of this method in the zebrafish (Danio rerio) embryo. Zebrafish serve as an ideal model species for this study, owing to their high fecundity and rapid exogenous development. Moreover, their redox biology closely mirrors humans – our data align with glutathione levels during human embryonic development.
Read the full study here in Redox Biology, an official journal of SfRBM: https://www.sciencedirect.com/science/article/pii/S2213231719302757
The organ-specific glutathione localization patterns we found lay the groundwork for determining discrete redox-sensitive temporal windows during embryogenesis. As illustrative examples of this, we tested two environmental toxicants – PFOS (a surfactant in non-stick cookware and firefighting foam) and MEHP (a bioactive metabolite of a ubiquitous plasticizer). We found that monochlorobimane responded in a dose-dependent, age and structure specific manner to both chemicals, indicating its broad application potential as an unbiased method to assess glutathione interruptions arising from developmental chemical exposures in live embryos.
— Published