Click here for the full July 2015 edition of SFRBM's DOT Newsletter
DOT: Tell us about your background and current passion in your professional life?
My father was an experimental physicist and I remember watching him assemble a hand blown glass vacuum apparatus to test the hypothesis that led to his graduate degree from the University of Vermont. My own first hands-on laboratory experience outside a classroom was at Argonne National Laboratory where I worked with Dr. Thomas M. Seed on low dose-rate gamma radiation-induced granulocytic leukemia in canines. This led to an interest in radiation biology and cancer and my matriculation to the University of Wisconsin-Madison where I performed graduate studies in the Human Oncology department under the mentorship of Dr. Kelly H. Clifton. My graduate training was in classical cellular radiation biology and carcinogenesis with a strong emphasis on organismal physiology and endocrinology. Since ~70% of the biological effects of gamma and x-ray irradiation are mediated through the radiolysis of water to generate oxygen free radicals, a natural interest in free radical processes in biology and medicine was forged from this background. I furthered my training at the post-doctoral level pursuing studies in molecular biology, carcinogenesis and redox-signaling at the University of Arizona Cancer Center in the laboratory of Dr. G. Tim Bowden. In 1993 I joined the University of Iowa and was fortunate to work as a colleague of Dr. Larry W. Oberley for 15 years until his untimely death in 2008. His inspiration helped crystalize for me a vision for how cellular redox metabolism communicates with epigenetic writer and eraser enzymes which when perturbed lead to aberrant epigenetic marks observed in tumor cells. I have pursued this general concept and many other side projects during my 23 year tenure at The University of Iowa. The current passion in my professional life is to watch the professional and career development of my numerous trainees from over the years unfold as they become successful in their own independent research careers and carry the torch of knowledge forward to the next generation.
 |
|
Rick Domann, Ph.D. |
DOT: Briefly describe your research interest and what is the most notable research achievement from your lab?
My research interests are in redox signaling, metabolism, cancer and epigenetics. I seek to understand how long-term perturbations to normal redox homeostasis such as those that occur in pathologies are drivers of an altered epigenetic landscape that changes phenotype without changing genotype. The most notable achievement from our lab has been the discovery that unfolded over the last five years which is that the SOD3 gene that encodes the extracellular superoxide dismutase is a potent metastasis suppressor gene and that its loss in several human epithelial cancers causes an acceleration of disease progression and a decrease in metastasis-free survival. This is in part due to the potent anti-inflammatory effects of EcSOD; its absence allows the proliferation of free radical reactions that degrade and destroy sensitive ECM components and accelerate invasion and metastasis.
DOT: Who has been your greatest teacher? What do you think the most important factors that shaped your career?
This is a difficult question to answer because there were and still are several very important mentors in my life, and it is nearly impossible to select the ONE that has had the most influence. I’d have to say that looking back at the direction of my work and the publications resulting over the last 30 years that my most influential teachers and mentors were Kelly Clifton and Larry Oberley.
DOT: In the current climate in which investigators are faced with decreased NIH funding for research and low morale, what is the best advice?
There is always room for optimism, and recent reports are that NIH may get a long-needed boost in support from Congress. Newt Gingrich recently wrote an Op-Ed in the New York Times (Apr 21, 2015) calling for another doubling of the NIH budget, and in mid-May the U.S. House of Representatives released a draft bill that aims to increase the NIH budget by $10 billion over 5 years. These shifting winds mean that investigators should stay tuned for additional evolving opportunities. Your Society leadership is trying to bring awareness to the field of redox biology and medicine at the NIH and other federal granting agencies by bringing scientific review administrators and program officers to the annual meetings to hold workshops for our members and highlight recent trends and funding opportunities in our field.
DOT: Being a mentor, you have shaped many students (graduate and postdoc) to enter academic and industry research, any tips how to shape individuals for these scientific fields?
There is no substitute for assiduity and a solid work ethic; being present is the greatest part of success. Beyond that, astute observational and critical thinking skills are required in a scientific profession of any kind. In addition, organizational, time management, and effective interpersonal communication are all key elements to success in science. I believe that proposals and reports need be compiled in a technically accurate manner and written in cogent prose in both academic and industry roles, so I think at least at the graduate training level there is little difference in how I would prepare them. At the post-doctoral level, a postdoc interested in industry may test the waters first by doing an industry post-doc or internship.
DOT: What do you think is the direction the Oxidative Stress field is heading?
Interesting question… I think that life is stress, especially life in an oxygen rich world. Thus all living organisms are in a non-steady state equilibrium that oscillates around various levels depending upon physical, biological, and environmental cues. A normal state of existence might be thought of as “eustress”, which is a normal and good part of life that is hormetic and that allows for rapid adaptive responses in either direction to maintain cell and tissue redox balance. Additionally, there is finally a realization in the community that redox sensors have been frequently misconstrued as antioxidant defenses when in many cases they are in fact signal transducing modules. All major disease of mankind possess an oxidative stress component to their pathologies, and so in order to cure these disease a deeper understanding of the redox signaling relays evolved in life in oxygen is needed.
DOT: And in the other direction, translational research.
Translational research utilizing basic knowledge learned from redox biology is growing exponentially. The role of free radicals and redox biology in inflammation, cancer, diabetes, cardiovascular and pulmonary diseases is well appreciated. Now, pharmacological control of certain of these systems is well in hand and drugs that mimic SOD activity are in clinical trials. Also, the paradoxical pro-oxidant effects of high dose vitamin C are making a big comeback in cancer clinics nationally with promising results using high C as an adjuvant for traditional therapies with no notable toxicity. Also, the role of redox couples that sense and signal oxidative stress, such as NADH/NAD+ are found to be vitally important for aging and cancer by altering the activity of sirtuin family enzymes, and oral nicotinamide was recently found to provide significant protection from non-melanoma skin cancer in high risk people. These are just a few examples of a growing list of medical applications forged from the furnace of free radical biology.
DOT: How has science/research changed during your life as a scientist?
I finished my BS in Biology in 1983, started grad school in 1984, and PCR was invented in 1985. That single method revolutionized experimental biology. In 1998 endothelial-derived relaxing factor was discovered to be nitric oxide which led to the Nobel Prize for scientists working in the field of free radical biology. I’ve always thought it was curious that the work on NO led to a prize, but the discovery of superoxide in biological tissues by Fridovich and McCord didn’t, especially since this was the seminal finding that gave birth to the field of free radical biology. The human genome initiative was completed by 2003; before that the GenBank was a fairly primitive and barely searchable database. The complete sequencing of many human genomes and the associated browser capability to navigate the human genome has changed everything about the future of medicine. Through that process, advances in DNA sequencing technology and speed have exceeded all expectations and has improved faster than Moore’s law. This is enabling an unprecedented examination of human microbiomes, genetic heterogeneity of cancer, even identifying heritable disease genes of embryos in utero from non-invasive blood tests of the mother. Finally, and directly relevant to mitochondrial redox biology, the United Kingdom recently approved the “three-parent baby”, which is aimed at eradicating familial mitochondrial diseases.
DOT: How important is the SFRBM conference to you and your trainees? Being elected as the president, what are the ideas are you planning for this society?
The annual SFRBM meeting has always been an extremely important meeting for me and my trainees. I have attended every meeting but one since 1994 and have usually travelled with a small entourage of trainees. The annual meeting has been important to me because it has allowed me to showcase my lab’s work in a receptive and like-minded audience. My trainees have been frequently selected for talks and awards and have benefited enormously from their sustained involvement in the Society. The Society is at a crossroads now, many members believe that it is time to re-brand and become more inclusive of all redox biology rather than exclusive to free radicals. Membership has been declining over the last few years and it is time to reinvigorate and re-energize the Society with an infusion of fresh blood that can most effectively be accomplished by broadening the scope and appeal of the Society to a wider scientific audience, one that would encompass and embrace all relevant redox biology in pathology and medicine. I have met personally with members of the membership committees and they are working with me on proactive strategies to not only retain existing members but also to recruit new members from all possible avenues. I believe that our new membership in FASEB will help in this regard, but also our Society would benefit from becoming more proactive at recruiting new members from like-minded scientists in other professional societies.
DOT: What are your hobbies outside the laboratory?
I enjoy reading, music, cooking, biking, traveling, hiking, fishing, & downhill skiing. I walk my dogs daily and always strive for at least ten thousand steps per day on the pedometer!
— Published
Category: SfRBM Member Profile
Click here to view the complete article in Redox Biology
Most of the SOD mimics thus far developed belong to the classes of Mn-(MnPs) and Fe porphyrins(FePs), Mn(III) salens, Mn(II) cyclic polyamines and metal salts. Due to their remarkable stability we have predominantly explored Mn porphyrins, aiming initially at mimicking kinetics and thermodynamics of the catalysis of O2− dismutation by SOD enzymes. Several MnPs are of potency similar to SOD enzymes. The in vivo bioavailability and toxicity of MnPs have been addressed also.
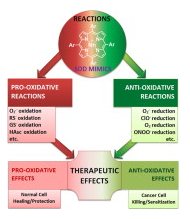 Numerous in vitro and in vivo studies indicate their impressive therapeutic efficacy. Increasing insight into complex cellular redox biology has been accompanied by increasing awareness of complex redox chemistry of MnPs. During O2− dismutation process, the most powerfulMn porphyrin-based SOD mimics reduce and oxidize O2− with close to identical rate constants. MnPs reduce and oxidize other reactive species also (none of them specific to MnPs), acting as reductants (antioxidant) and pro-oxidants.
Numerous in vitro and in vivo studies indicate their impressive therapeutic efficacy. Increasing insight into complex cellular redox biology has been accompanied by increasing awareness of complex redox chemistry of MnPs. During O2− dismutation process, the most powerfulMn porphyrin-based SOD mimics reduce and oxidize O2− with close to identical rate constants. MnPs reduce and oxidize other reactive species also (none of them specific to MnPs), acting as reductants (antioxidant) and pro-oxidants.
Distinction must be made between the type of reactions of MnPs and the favorable therapeutic effects we observe; the latter may be of either anti- or pro-oxidative nature. H2O2/MnP mediated oxidation of protein thiols and its impact on cellular transcription seems to dominate redox biology of MnPs. It has been thus far demonstrated that the ability of MnPs to catalyze O2−dismutation parallels all other reactivities (such as ONOO− reduction) and in turn their therapeutic efficacies.
Assuming that all diseases have in common the perturbation of cellular redox environment, developing SOD mimics still seems to be the appropriate strategy for the design of potent redox-active therapeutics.
— Published
Categories: Education, Redox Biology