Part 1 of 4 on “Exploring Careers in Industry” by SfRBM Nominations/Leadership Development Chair Anne Diers, Ph.D. (Dr. Diers on LinkedIn: https://www.linkedin.com/in/annerdiers/)
This might sound obvious, but “Industry” is a really broad term – so when you think of a job in industry, you’re actually talking about many huge sectors of science. The available jobs will be very different based on what you’re looking for. So you need to ask yourself a few questions to narrow in on the type of position you’re interested in. Do you want to be bench-facing? Would you like to manage a group of scientists? Do you love writing about science? Do your interests lie in product development, management, pharma, bio-tech, or instrumentation? These are obviously critical questions and you have to answer them for yourself. If the answers aren’t obvious, spend some time exploring different areas of non-academic science. myIDP at Science Careers is a very helpful tool in this respect  (myidp.sciencecareers.org/). In fact, there is an incredible wealth of information on Science Careers itself. Another good way to get a handle on what you're looking for is to talk to people doing these various jobs. Talk to your sales reps, see who they know and if they can connect you to people, set-up informational interviews to find out what people actually do in their jobs.
(myidp.sciencecareers.org/). In fact, there is an incredible wealth of information on Science Careers itself. Another good way to get a handle on what you're looking for is to talk to people doing these various jobs. Talk to your sales reps, see who they know and if they can connect you to people, set-up informational interviews to find out what people actually do in their jobs.
Looking at job sites for current openings can also help you refine your idea of an ideal position. First, you’ll see what opportunities are already open. But second, and more importantly, you’ll get an idea for what job titles match the job you want to do (and for which you have the necessary experience). You’ll start to develop a feeling for whether or not you want to be a Field Applications Scientist, or a Program Manager, or whatever, based on those descriptions. Then, when you find yourself in conversation with your sales reps, colleagues, or faculty, you can say to them “I’m looking for an entry-level [ideal position for you]. Have you heard of any openings for this type of job at [their illustrious company]?” This instantly gives you some credibility… and rightfully so. It means that you have done your homework, and you have a real idea of what you want to do.
In part 2 of this series, Dr. Anne Diers will explore the topic “Where do I want to work… and who already works there?” In her series “Exploring Careers in Industry.”
— Published
Categories: Education, SfRBM Trainee Council
By: Eugenio Barone and Marzia Perluigi
Read the full articles in Free Radical Biology and Medicine here: Barone E et al. Free Radic Biol Med, 111:262-269, 2017, Barone E et al. Free Radic Biol Med, 114:84-93, 2018
Accumulation of oxidative damage is a common feature of neurodegeneration that, together with mitochondrial dysfunction, point to the fact that reactive oxygen species are major contributors to loss of neuronal homeostasis and cell death. Among several targets of oxidative stress, free radical-mediated damage to proteins is particularly important in aging and age-related neurodegenerative diseases. In the majority of cases, oxidative stress mediated post-translational modifications cause non-reversible modifications of protein structure that consistently lead to impaired function.
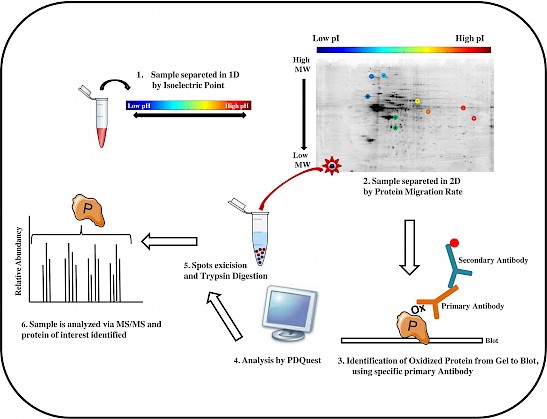
Figure: The workflow of a redox proteomics analysis is showed in six principal steps.
Redox proteomics methods are powerful tools to unravel the complexity of neurodegeneration, by identifying brain proteins with oxidative post-translational modifications that are detrimental for protein function and pathogenesis (Butterfield DA et al. Biochem J, 463:177-89, 2014). Our published studies show evidence of impaired pathways linked to oxidative stress possibly involved in the neurodegenerative process leading to the development of Alzheimer-like dementia (Di Domenico F et al. Antioxid Redox Signal, 26:364-387, 2017). Recently, we also focused on dysregulated pathways underlying neurodegeneration in aging adults with Down syndrome (DS) (Barone E et al. Free Radic Biol Med, 111:262-269, 2017). Interestingly, DS individuals by the age of 40ys are at increased risk to develop Alzheimer disease (AD) like dementia.
Results obtained by the analysis of human specimens and studies from mouse and cellular models of the disease evince a molecular link between protein oxidation/aggregation, the integrity of protein quality control system [proteasome, unfolded protein response (UPR) and autophagy], dysfunction of energy metabolism and neurodegeneration (Di Domenico F et al. Antioxid Redox Signal, 26:364-387, 2017). Many common pathological hallmarks exist between DS and AD including deposition of b-amyloid plaques, Tau-based neurofibrillary tangles, increased oxidative damage, and impaired mitochondrial function, among others. Intriguingly, we propose that all these processes seem to be joined by a “leitmotif” – oxidative stress - since they are all the cause and/or the consequence of increased free radical burden. If low amounts of reactive oxygen species (ROS) can activate the protective cellular apparatus such as the antioxidant and heat shock responses, cell cycle regulation, DNA repair, UPR and autophagy, then chronic exposure to ROS causes irreversible damage to all intracellular macromolecules (Barone E et al. Free Radic Biol Med, 111:262-269, 2017). Among these, protein oxidation impairs multiple cellular functions by a largely irreversible process that results in altered, mostly reduced, protein activity. It is likely that stressed neurons have the challenge of increasing loads of oxidatively damaged proteins, which overwhelm the ability of the proteostasis network. This, in turn, promotes further accumulation of damaged proteins, increasingly prone to aggregation, ultimately resulting in neuronal death. Alteration of protein homeostasis coupled with increasing demand for protein degradation, and reduced ATP production may produce a vicious cycle that may accelerate the neurodegenerative process (Barone E et al. Free Radic Biol Med, 111:262-269, 2017, Barone E et al. Free Radic Biol Med, 114:84-93, 2018).Therapeutic strategies aimed at preventing/reducing multiple components of processes leading to accumulation of oxidative damage will be critical in future studies.
Laboratory of Redox Biochemistry in Neuroscience, Department of Biochemistry, Sapienza University of Rome, Email: Eugenio.barone@uniroma1.it
— Published
Category: Redox Biology
By: Matthew Randall, Ph.D. ETH Zürich, & Niki Ubags, Ph.D., CHUV, Lausanne
Exploration is the crux of science, whether it is that of visible or invisible phenomena. Without the exploration of faraway lands, Darwin’s theory of evolution would not exist. However, today’s technology provides scientists with more information than ever, so is this type of exploration really still needed? We can explore places or accomplish many things without leaving the comfort of our home or office. So, why cross an ocean when you can simply collaborate through a video chat with scientists from around the globe?
Crossing the ocean for research is not limited to simply acquiring data or becoming knowledgeable about a specific subject matter. By living in a different environment, different culture, language, et cetera you may find yourself starting to think differently about the world around you and the way you conduct science or think about new scientific questions. In a sense, physical exploration brings mental exploration.
How does physically being somewhere else benefit you in your scientific career?
When you find yourself dropped into a strange new place, all of your senses become heightened and you first notice all of the differences (both cultural and social). Even the simplest of things become interesting, whereas the same things commonly encountered at home were too mundane to see from a different perspective. This outsider’s perspective is the first benefit to you. The research you would otherwise perform routinely will come into a new light and not only will others have the opportunity to help you view your projects differently, but you yourself begin to think differently. For example, a year ago I moved to Switzerland and joined a laboratory in the Institute for Biomechanics at ETH Zürich. Most of my colleagues are researching processes that range from mechanobiology signaling pathways to computer-generated simulations of bone fracture. Now, as a redox biologist, not only do I have the opportunity to discuss my research with people who approach problems in a different way, but I also begin to view my own research from this outside perspective. Moreover, I have begun to integrate their knowledge and thinking into my work and approach my questions from a very different perspective than I had before.
When you place yourself in someone else’s shoes, and begin to view things from another perspective, you may also begin to - in essence - find yourself. This active mindfulness, as I call it, is critical for navigating not only life, but also career and research. What do I want? How should I get there? These are the questions that you will find yourself asking. Stepping out of your own shoes and looking at how you navigate in a new place with new people and situations, you come to understand your own preferences as well as how you feel in certain situations. Since people in other parts of the world communicate differently about what they want and how they feel, you face the challenge to understand not only how you express yourself, but also how others express themselves toward you. The ability to know yourself as well as understand others is certainly an earned skill of working internationally.
Every country, every institution, and every lab functions differently, therefore the best way to succeed in a new place is to adapt. When you begin to see yourself as a part of the world in which you once felt foreign, you might realize that you are capable of adaptation, even on a short-time scale. You are capable of using tools and skills that you had not known how to use before. The ability to adapt to not only your physical environment but also the political and research environments of a new institution promotes your flexibility and creativity in your character as well as in your research.
Now you may be thinking… how long do I have to be abroad to become “enlightened.” It is not necessary to move abroad permanently to benefit from these experiences. In the case for Darwin, it was only after his return to England from the Galapagos, while contemplating his drawings, that he would develop his theory of evolution. In a similar way, returning home with a new perspective on your own research and life may lead to the greatest of discoveries of yourself and your career.
— Published
By: Ines Batinic-Haberle and Ivan Spasojevic
After 20 years of bench and preclinical work on Mn porphyrins (MnPs), we are thrilled to see that our compounds  have finally reached maturity and entered four clinical trials. We have first designed, synthesized and characterized numerous MnPs, FePs and other metal complexes, which allowed us to establish structure-activity relationship for their SOD-like activities and identify cationic charges in ortho nitrogen positions of peripheral pyridyl groups of MnPs as a key property that enables their high ability to catalyze O2.- dismutation.
have finally reached maturity and entered four clinical trials. We have first designed, synthesized and characterized numerous MnPs, FePs and other metal complexes, which allowed us to establish structure-activity relationship for their SOD-like activities and identify cationic charges in ortho nitrogen positions of peripheral pyridyl groups of MnPs as a key property that enables their high ability to catalyze O2.- dismutation.
Since then, three lead drugs have been most frequently studied: MnTE-2-PyP5+(AEOL10113, BMX-010), MnTnHex-2-PyP5+ and MnTnBuOE-2-PyP5+(BMX-001) (Figure, R, ortho pyridyl substituents are ethyl, n-hexyl and butoxyethyl, respectively).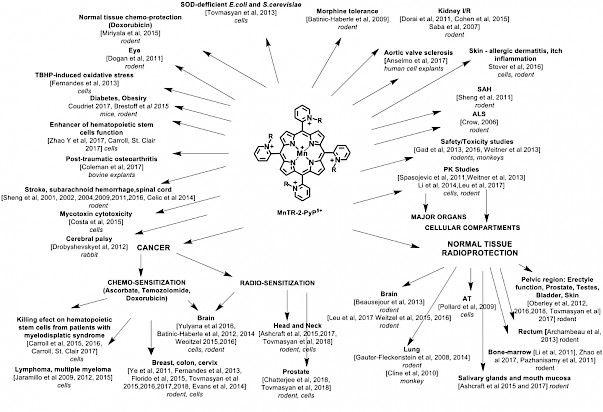 BMX-001 is now tested on suppression of normal tissue injuries in three clinical trials concerning the treatment of various cancers (submitted for publication). Originally designed as SOD-mimics, the biologically compatible reduction potential of Mn in MnP enables broad interactions with other species and redox signaling pathways. In collaboration with Tome’s group, we demonstrated catalysis of S-glutathionylation of Cys residues of NF-кB by MnP/H2O2/GSH, leading to NF-кB inactivation. In addition to NF-кB, S-glutathionylation of Cys was also demonstrated for signaling proteins in our redox proteomics study on 4T1 breast cancer cells treated with the MnP/ascorbate source of H2O2: p38MAPK, protein phosphatase 2A, Keap1, peroxiredoxins 5 and 6, glutaredoxin 3 and 5 and thioredoxin 1, etc.
BMX-001 is now tested on suppression of normal tissue injuries in three clinical trials concerning the treatment of various cancers (submitted for publication). Originally designed as SOD-mimics, the biologically compatible reduction potential of Mn in MnP enables broad interactions with other species and redox signaling pathways. In collaboration with Tome’s group, we demonstrated catalysis of S-glutathionylation of Cys residues of NF-кB by MnP/H2O2/GSH, leading to NF-кB inactivation. In addition to NF-кB, S-glutathionylation of Cys was also demonstrated for signaling proteins in our redox proteomics study on 4T1 breast cancer cells treated with the MnP/ascorbate source of H2O2: p38MAPK, protein phosphatase 2A, Keap1, peroxiredoxins 5 and 6, glutaredoxin 3 and 5 and thioredoxin 1, etc.
Most recently, in a comprehensive mouse breast cancer cellular and mouse study, we explored cationic MnPs (BMX-001, BMX-010), GC4403 (analog of GC4419), Mn(III) salen (EUK-8) and a porphyrin-based but not an SOD mimic, yet frequently studied, anionic MnTBAP3-. Only cationic MnPs, which catalyze ascorbate oxidation and protein S-glutathionylation, radiosensitize breast cancer to radiation, and ascorbate enhances such effect (Tovmasyan et al, Antioxid Redox Signal. 2018 Feb 1. doi: 10.1089/ars.2017.7218). Altogether, our studies show that S-glutathionylation of Cys residues is a major mechanism of action of MnPs. Multiple studies by us and others have shown that the more potent the SOD mimic is, the more reactive it is and the larger are its beneficial therapeutic effects whereby it protects normal tissue from oxidative stress injury while suppresses tumor growth (Figure).
Our pharmacokinetic and efficacy studies showed that differential effects originate from distinct partition of MnP in tissues as well as from tissue-specific redox environments. While therapeutic effects are those commonly associated with antioxidants, the mechanism of action associated with our MnPs is pro-oxidative – we thus tend to call such compounds more correctly redox-active therapeutics than antioxidants.
Departments of Radiation Oncology1 and Medicine2, 3PK/PD Pharmaceutical Shared Resource, Duke Cancer Institute, Duke University School of Medicine, Durham, NC 27707, USA
— Published
By Dr. Marcus S. Cooke, Ph.D., FRCPath of Florida International University
Luke Skywalker: “I won't fail you! I'm not afraid.”
Yoda: “Oh! You will be. You will be.”
Similar to Luke’s training by Yoda, the PhD is like an apprenticeship, students are mentored and supported throughout, but this is gradually withdrawn, as they become more accomplished, culminating with being an expert in the field, and an independent researcher. On this basis, selection of the right supervisor for you is as vital as selecting the right project area. The successful pursuit of a doctoral degree is a two-way street that requires good alignment between your objectives and those of your supervisor, as well as your progressive capacity to deal with failure and rejection (it’s a fact of life for researchers, I’m afraid). 
A PhD requires fulltime dedication to the project. Before considering undertaking a PhD a prospective student must be willing to devote themselves to the project, understanding that experiments sometimes require working outside business hours. You may possess a great deal of practical experience, but this experience by itself is not sufficient to earn the degree. The project fails or succeeds based on the effort, and ideas from the student, guided and supported by the supervisor. In order to have the required level of commitment, the chosen project on which you work must really excite you – this will provide the motivation to keep going, particularly when experiments prove challenging (i.e. techniques stop working for no apparent no reason!). There will be lows, but there will also be highs, for example when your abstract gets accepted for an international meeting, or your first manuscript gets accepted (it feels good whether it’s your first or 81st). This strengthening of your character will surely enrich your career and life.
The kinds of wet lab projects offered in the broad area of Biomedical Sciences do not readily lend themselves to a part-time degree, but that is not to say it is not possible. A PhD in the Biomedical Sciences means 3-4+ years of intensive research in the lab – sounds like plenty of time, right? But the time flies by in which you must have generated a significant amount of novel data, addressing an important research question. Also, don’t think that a PhD is simply an extension of a Master’s degree, only longer. It is completely different, with objectives, timelines and expectations of progress toward scientific independence that are not seen at the Master’s level. 
Good organization of personal time will be needed, such that you can improve skills that are just as important as your experimental work. This includes: keeping up with the literature about the subject, improving scientific writing, cultivating attention to detail, attending seminars and workshops in and outside your institution, and very importantly, mastering your own oral communication skills. Most institutions demand a minimum of two original data manuscripts, and a review article to submit towards your PhD. These will need to survive the scrutiny of peer review, which will train your capacity to receive criticism and to work proactively on improving the quality of your research.
A huge amount of self-motivation is required to withstand the intrinsic challenges of developing a scientific project toward thinking independently. The structured, taught element of the PhD is minor, in comparison to the time spent in the lab, and just provides a general grounding to get improve your knowledge base. The real learning and training comes in the lab where research skills are learnt firsthand, and tested on a regular basis through the experiments required to test your hypothesis.
Still wanting to do a PhD in the Biomedical Sciences? Get in touch with prospective mentors, discuss with them theirs and your research and career expectations, and go for it! If you are intrinsically thrilled to investigate Nature, you will not be hampered by the high level of commitment required in this career path. With the right PhD training (irrespective of the discipline), you will be well prepared for a wide variety of career options, though giving you skills which allow you to think, speak, write, and problem-solve in a particular way that makes you an asset to any organization.
— Published
Categories: Research, Free Radical Biology and Medicine, SfRBM Trainee Council, Redox Biology